Controversy shadows Chinese vaccine maker Sinovac
FILE PHOTO: CHINESE EMBASSY
MANILA, Philippines—Controversy continued to hound the Chinese pharmaceutical company Sinovac, maker of the coronavirus vaccine Coronavac.
A report about scores of Indonesian health workers getting infected despite having received Coronavac injections had drawn concern among countries relying on the Chinese vaccine.
Last week, Indonesia’s health officials revealed that over 350 doctors and medical workers contracted SARS Cov2, the virus that causes COVID-19, despite getting injected with Coronavac.
Scores of health care workers in Indonesia were hospitalized with high fever and low oxygen saturation levels. Most had no symptoms and were on self-quarantine at home.
The report raised concern because it came as the district of Kudus in Central Java reported an outbreak of SARS Cov2 infections believed to be caused by the more transmissible delta variant.
Article continues after this advertisementHundreds of vaccinated Indonesian health workers get COVID-19, dozens in hospital
Article continues after this advertisementWhile public health experts in Indonesia acknowledged Coronavac’s impact on reducing COVID deaths among health care workers, recent hospitalizations following inoculation remained a cause for concern, especially on whether the Sinovac product was really effective.
How effective is Coronavac against COVID-19?
Clinical trials have been conducted in several countries to measure the efficacy of Sinovac’s coronavirus vaccines.
INFOGRAPHIC BY ED LUSTAN
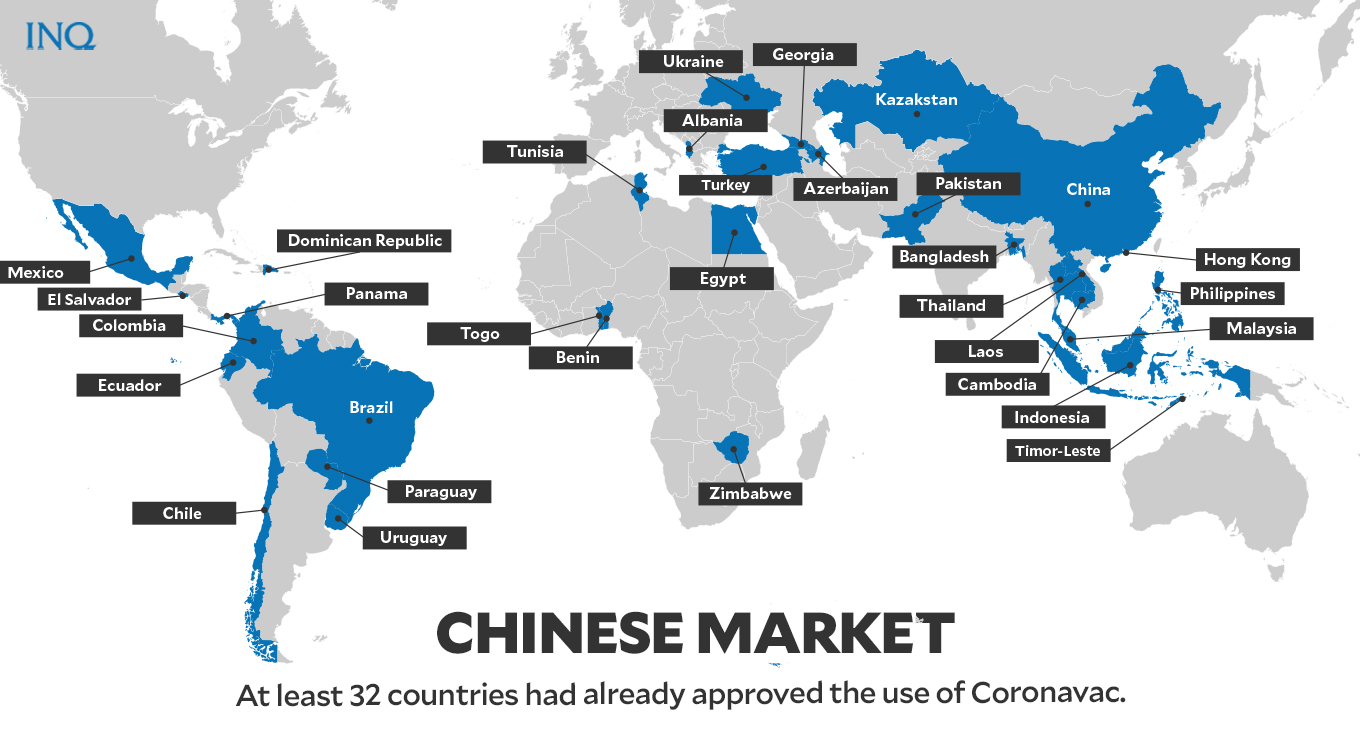
According to COVID-19 Vaccine Tracker, at least 32 countries had already approved Coronavac for emergency use:
- Albania
- Azerbaijan
- Bangladesh
- Benin
- Brazil
- Cambodia
- Chile
- China
- Colombia
- Dominican Republic
- Ecuador
- Egypt
- El Salvador
- Georgia
- Hong Kong
- Indonesia
- Kazakstan
- Laos
- Malaysia
- Mexico
- Pakistan
- Panama
- Paraguay
- Philippines
- Thailand
- Timor-Leste
- Togo
- Tunisia
- Turkey
- Ukraine
- Uruguay
- Zimbabwe
The different trials, however, have produced varying efficacy data in past months.

INFOGRAPHIC BY ED LUSTAN
Indonesia, the first country to give Coronavac an emergency use approval outside China, reported last January that the vaccine was 65 percent effective after phase 3 clinical trials.
Indonesia started its nationwide rollout of Coronavac last Jan. 4, giving the first shots to health and frontline workers.
Indonesia starts nationwide Sinovac COVID-19 vaccine distribution
Last May, Indonesia released another promising data detailing Coronavac’s efficacy.
The study conducted by Indonesia’s health ministry showed that the vaccine was 98 percent effective at preventing death and 96 percent effective at preventing hospitalization.
The results were based on data from 128,920 health care workers in Jakarta who were inoculated between January 13 to March 18.
The late-stage trial also showed that the vaccine prevented symptomatic COVID-19 in 94 percent of the group.
Turkey, which was among the first to release data on Coronavac’s efficacy, said last March that the vaccine was 83.5 percent effective against coronavirus.
This was lower compared to its initial findings which suggested an efficacy rate of 91.25 last December. However, researchers maintained that the vaccine was still 100 percent effective at preventing severe cases of COVID-19 and hospitalization.
The most recent data were based on late-stage trials among 10,216 participants—6,648 of whom received jabs as part of the study which began late September.
Brazil’s clinical trials, on the other hand, resulted in an efficacy rate of 50.4 percent against symptomatic infections—lower than the regulatory approval and below the initially announced rate of 78 percent.
New Brazil data shows disappointing 50.4% efficacy for China’s Sinovac vaccine
However, the World Health Organization (WHO) said on its website that the vaccine—based on the phase 3 trials in Brazil—was 100 percent effective against severe COVID infections and 100 percent effective in preventing hospitalization starting 14 days after the second dose.
10 million test subjects in Chile
Sinovac’s vaccine was found to be 67 percent effective in preventing symptomatic infection, according to a huge real-world study in Chile.
The Chilean government in April said the vaccine was 85 percent effective in preventing hospitalizations and 80 percent effective in preventing deaths.
Game changer: Chile hails Sinovac’s 67% effectiveness in study
The study examined over 10 million people in Chile’s public health system between February and April. A comparison was made between people who have not yet received vaccine doses, individuals 14 days or more after receiving one dose, and more than 14 days after receiving a second dose.
Most recently, Uruguay found that the vaccine was 61 percent effective at preventing infections.
It was also 92 percent effective in preventing intensive care admissions and has reduced deaths by 95 percent.
The study was conducted among 795,684 health workers and the general population between 18-69 years old.
The results were based on the participants’ condition at least 14 days after their second dose.
Varying success rate
According to experts, the different efficacy rates can be attributed to many factors including:
- trial size
- target population or groups (health care workers, elderly, persons with comorbidity, etc.)
- duration of the post-vaccination observation
- COVID-19 situation in each site (number of active cases and deaths)
What’s behind varying efficacy data for Sinovac’s COVID vaccine?
But Sinovac itself has yet to release its data on the effectiveness of the Coronavac against new SARS Cov2 variants.
What recipient countries say
Last week, Singapore allowed several private health clinics to administer vaccines manufactured by China’s Sinovac.
However, despite the seemingly strong demand for Coronavac doses, the government still cast doubts over the vaccine’s efficacy.
“It does give the impression that the efficacy of different vaccines will vary quite significantly,” Singapore’s director of medical services Kenneth Mark told the New York Times last Friday (June 18), citing the incident in Indonesia.
Mark said he was worried over reported cases where individuals still became sick even after receiving jabs of Sinovac’s vaccine.
In the Philippines, health officials maintained that the Chinese firm’s COVID vaccine remained effective against severe infections.
Sinovac vaccine remains effective vs severe COVID-19, says DOH
“Real-world studies showed that all vaccines, including those being used in the country, are all effective. It means it can prevent severe infections and hospitalization,” said Health Undersecretary Maria Rosario Vergeire in Filipino.
Vergeire added that more information is still needed about what happened to Indonesia’s health workers who received Coronavac shots.
She also advised the public to be cautious in interpreting similar information and reports.
“Looking at that article, we need to be very cautious because the people might get scared,” Vergeire said. “That article did not detail whether the affected health care workers were fully vaccinated or they just received one dose,” she said.
“Even our experts have reviewed this and we are still looking for other data to complete our analysis based on their report,” she continued.
The DOH urged the public to get vaccinated and not be doubtful of vaccines approved by the government.
The Philippines has so far received a total of 9 million COVID-19 vaccine doses by Sinovac—a million of which were donated by China.
Data on clinical studies of Coronavac showed these results per country:
- December 2020 in Turkey. Coronavac efficacy rate was 91.25 percent according to results of phase 3 trials involving 7,000 volunteers.
- Jan. 7 and Jan. 12, 2021 in Brazil. First efficacy rate was 78 percent and second was 50.4 percent. At least 13,000 people took part in trials with results released on Jan. 7.
- Jan. 11, 2021 and May 12, 2021 in Indonesia. Coronavac efficacy was 65.3 percent in phase 3 trials based on Jan. 11 results. Efficacy was 98 percent in results on May 12.
- March 4, 2021 in Turkey. Efficacy rate was 83.5 percent in phase 3 trial involving 10,216 participants
- April 17, 2021 in Chile. Efficacy rate was 67 percent in trials that involved 10.5 million people.
- June 9, 2021 in Uruguay. Efficacy rate was 61 percent in trials involving 795,684 people
TSB
For more news about the novel coronavirus click here.
What you need to know about Coronavirus.
For more information on COVID-19, call the DOH Hotline: (02) 86517800 local 1149/1150.
The Inquirer Foundation supports our healthcare frontliners and is still accepting cash donations to be deposited at Banco de Oro (BDO) current account #007960018860 or donate through PayMaya using this link.