Blank space left in 2022 PH budget: Funds for free COVID tests
MANILA, Philippines—Amid the alarming spike of COVID-19 cases in the country following the holiday celebrations, a lawmaker pointed out something very glaring but overlooked in the P5.024 trillion 2022 national budget—there were no funds allotted for free mass testing.
In a statement on Tuesday (Jan. 4), House Deputy Minority Leader and Bayan Muna Rep. Carlos Isagani Zarate said this year’s budget is “not designed to save lives” as it gave priority to funding the procurement of arms and military equipment.
“The national budget is not designed to save the lives of Filipinos but to pander to the security sector’s militarist whims and at the same time, enrich the makers of arms and war materiel,” Zarate said.
“There are no money for free mass testing for vulnerable persons, essential workers, and employees, but there is money to buy missiles, helicopters and war materiel,” he added.
The lawmaker also lamented cuts in the budgets for the Philippine General Hospital (PGH) and the Food and Drug Administration’s (FDA) in the 2022 General Appropriations Act (GAA), the law passed by Congress to finance government operations.
Article continues after this advertisementHe also revealed the lack of funds for the P45.368 billion budget for the procurement of COVID vaccine booster shots, which he said remained under unprogrammed appropriations.
Article continues after this advertisementIn contrast, the Department of National Defense (DND), Philippine National Police (PNP) and the AFP Modernization Program all got additional funding.
“In the twilight of the Duterte administration, it is prioritizing and splurging billions of pesos on weapons, yet, it is claiming that it has no money for free, systematic widespread testing,” said Zarate.
Cost of COVID-19 tests
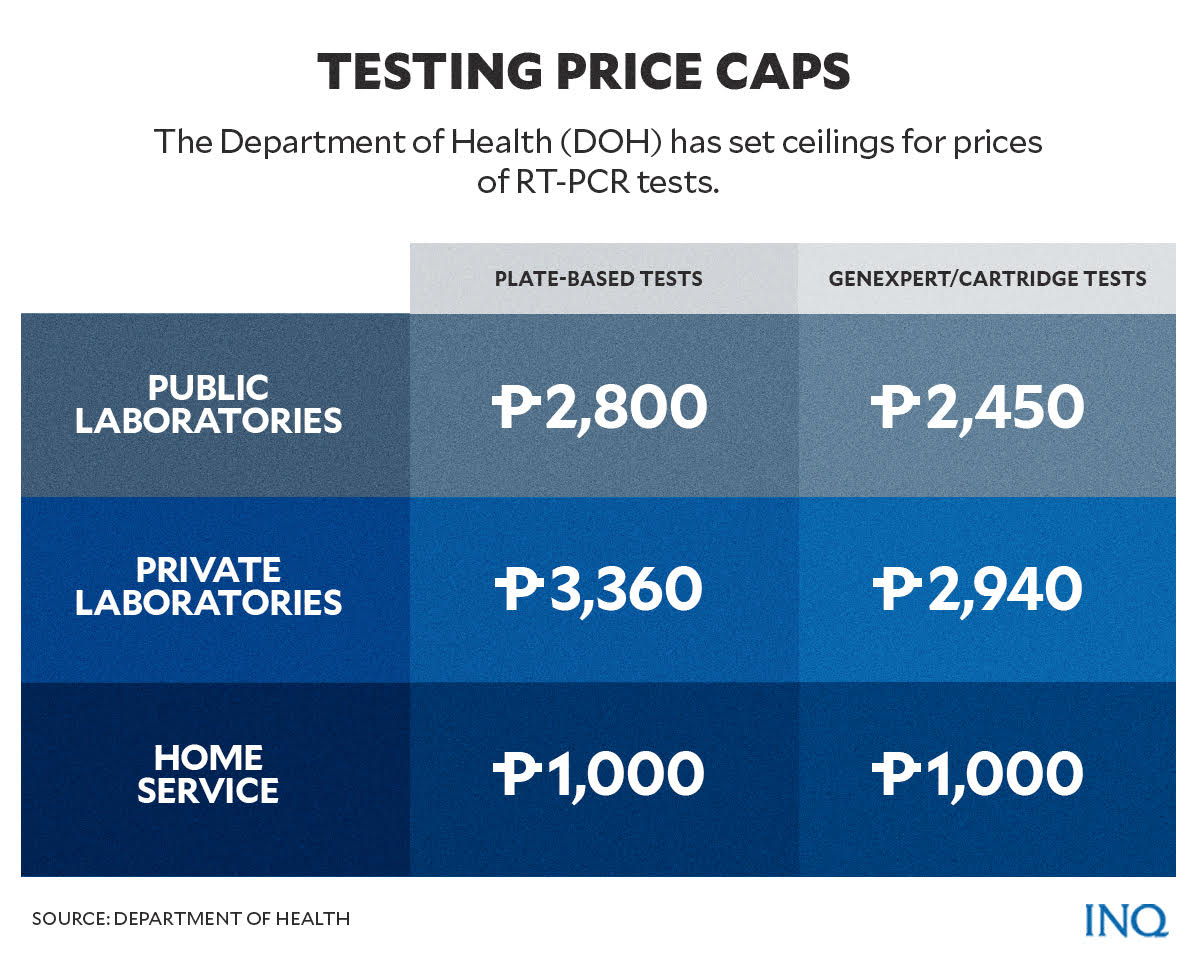
In August last year, the Department of Health (DOH) released Circular No. 2021-0374, which adjusted the price cap for Reverse Transcription Polymerase Chain Reaction (RT-PCR) tests in the country.
According to DOH, it has made COVID-19 testing more affordable to the public amid rising cases of coronavirus infections, as well as the then-emerging threat of the highly transmissible Delta variant.
Since Sept. 6, 2021, the price caps for RT-PCR tests were:
- For public laboratories: P2,800 for plate-based and P2,450 for GeneXpert or cartridge-based
- For private laboratories: P3,360 for plate-based and P2,940 for GeneXpert or cartridge-based
- Home service: P1,000 for plate-based and GeneXpert or cartridge-based
READ: Surviving COVID-19: A costly struggle
The health department noted that laboratories, health facilities, and other testing centers may offer prices lower than the prescribed price cap for RT-PCR testing and home service fee, provided that quality of service is maintained.
However, manufacturers, distributors, and testing facilities are not allowed to charge higher than the prescribed price caps.
The DOH also stated in the circular that senior citizen and persons with disability (PWD) discounts are applicable to the total amount of services rendered for home service.
Outside of government, two types of tests are offered by the Philippine Red Cross (PRC)—the RT-PCR test, which was considered to be the “gold standard” in testing, and the cheaper and less invasive saliva-based COVID test.
READ: EXPLAINER: What is saliva-based Covid-19 test and where to get it
According to PCR chair Richard Gordon, prices of RT-PCR and saliva tests became 25 percent cheaper since September last year.
READ: Red Cross’ saliva, swab test price now 25% down starting Sept. 4
Below are the current prices of COVID tests offered by the PRC:
- RT-PCR test: P2,800
- Saliva test: P1,500
PhilHealth coverage
Last month, the Philippine Health Insurance Corp. (PhilHealth) announced its new benefit package rates for COVID-19 testing, through Circular No. 2021-0021, which took effect on Nov. 30, 2021.
According to the state insurer, its revised benefit packages were based on the revised costing estimates and rates prescribed by the DOH and the Department of Trade and Industry (DTI).
The revised benefit package rates are as follows (package code, condition for payment, services covered by PhilHealth, and package amount):
Package rates for plate-based RT-PCR test
- C19T1: All services and supplies for the testing are procured and provided by the testing laboratory.
Package covers complete services or minimum standards.
Package amount: P2,800 - C19T2: Test kits are donated to the testing laboratory.
Package covers screening, specimen collection and handling, the conduct of RT-PCR testing, and analysis of results.
Package amount: P1,200 - C19T3: Test kits are donated to the testing laboratory, while the cost of running the laboratory and the RT-PCR machine for testing are subsidized by the government.
Package covers: screening, specimen collection and handling.
Package amount: P800
Package rates for cartridge-based PCR test
- C19X1: All services and supplies for the testing are procured and provided by the testing laboratory.
Package covers complete services or minimum standards.
Package amount: P2,450 - C19X2: PCR cartridges are donated to the testing laboratory.
Package covers screening, specimen collection and handling, the conduct of cartridge-based PCR testing, and analysis of the results.
Package amount: P700 - C19X3: PCR cartridges are donated to the testing facility, while the cost of running the cartridge-based PCR test is subsidized by the government.
Package covers screening, specimen collection and handling, and the facility costs for the staff time and PPE (personal protective equipment).
Package amount: P500
The state insurer said the circular applies to PhilHealth beneficiaries tested for SARS-CoV-2, the virus that causes COVID-19, through RT-PCR (whether cartridge-based or plate-based) “as prescribed by the DOH, and to DOH-licensed PhilHealth-accredited SARS-CoV-2 testing laboratories.”
Those who are not registered in PhilHealth “shall automatically be covered, provided that they complete member registration upon availment of the benefit package.”
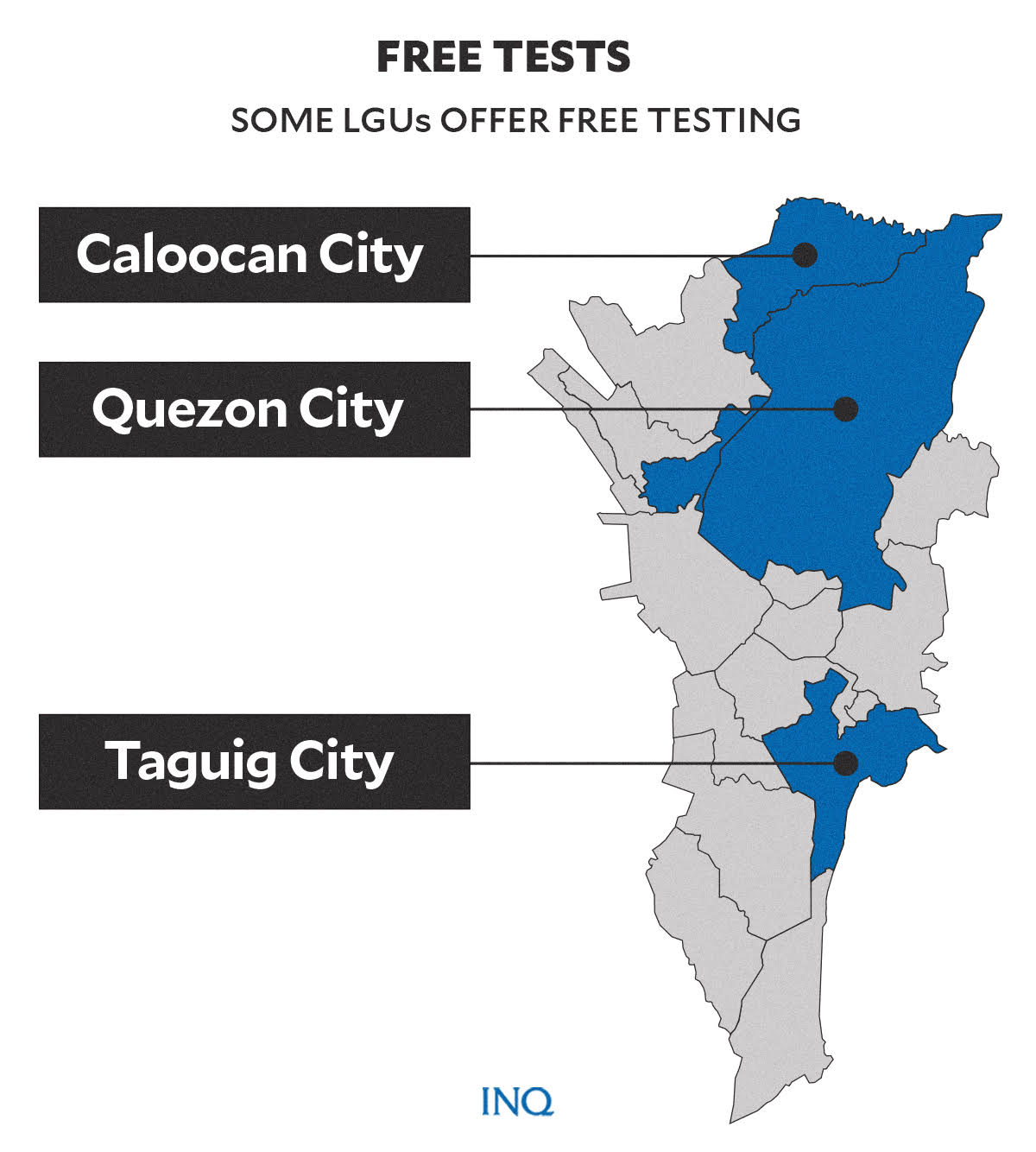
LGUs with free, cheaper options
To help their constituents avail themselves of tests amid the recently increasing cases of COVID-19, some local government units (LGUs) in Metro Manila announced that they will offer free COVID testing to their residents.
On Jan. 4, Caloocan City Mayor Oscar Malapitan announced that there will be a free mobile antigen swab testing in several barangays from 8:30 a.m. to 11 a.m. starting Jan. 4 to Jan. 7.
Malapitan said that aside from the mobile testing, the city government continues to offer free RT-PCR testing at the Caloocan People’s Park at Caloocan Sports Complex every weekday.
In Quezon City, the Quezon City Epidemiology & Disease Surveillance (CESU) continues to offer free swab tests for residents who are experiencing symptoms of COVID-19 at the city’s community-based testing sites (CBTs).
Among those given priority in CBTs included close contacts of COVID-positive individuals, those who are experiencing COVID-19 symptoms, pregnant women, and those who need to be tested for medical operations.
On Jan. 1, the city government of Taguig announced that citizens can schedule their free RT-PCR test through the website: www.trace.taguig.gov.ph.
In a Facebook announcement, the Taguig City government said its City Epidemiology and Disease Surveillance Unit (CEDSU) and Barangay Health Centers are conducting free PCR testing from Monday to Friday.
Regular drive-through tests are also available every Wednesday at BGC and every Friday at Lakeshore, the city government announced.
“The city also launched Park ‘N Test which will be accessible every Monday at the Vista Mall Parking Building,” it said.
“Another way to be tested is through the Mobile Testing Truck making its rounds in the city,” it added.
Antigen self-test kits
On Jan. 5, the DOH said that together with the FDA and the Research Institute for Tropical Medicine (RITM), it will study whether to recommend the use of antigen self-test kits to detect the COVID virus.
READ: DOH studying use of antigen self-test kits
“We just need to ensure that there is no misuse of the self-test antigen because we should use the right test at the right time, there should be appropriate use,” said Health Undersecretary Ma. Rosario Vergeire.
“We are studying how we can use this in a way that there would be no inaccurate results,” she added.
More COVID, more profit
Last year, US business magazine Forbes published an article—“Meet The 40 New Billionaires Who Got Rich Fighting Covid-19”—which listed companies and individuals who recently got rich through big companies, like Moderna and BioNTech, which supply key COVID supplies, including vaccines.
Some of the newcomers on the billionaires’ list grew their fortunes on manufacturing and supplying PPEs, antibody treatments, software used to schedule vaccination campaigns, and diagnostic tests.
- Among those who entered the list of the world’s wealthiest people through COVID-19 tests were:
- Dai Lizhong of Sansure Biotech—net worth: $2.4 billion
- Sergio Stevanato of the Stevanato Group-net worth: $1.9 billion
- Ulrike Baro of Sartorius AG-net worth: $1.5 billion
- Arvind Lal of Dr. Lal Pathlabs-net worth: $1.5 billion
- August Troendle of Medpace-net worth: $1.5 billion
- Liang Yaoming of Guangzhou Kingmed Diagnostics Group-net worth: $1.4 billion
- Alan Miller and family of Universal Health Services-net worth: $1.3 billion
- Rao Wei and family of Shenzhen New Industries Biological Engineering-net worth: $1.2 billion
- Li Wenmei and family of Guangzhou Wondfo Biotech-net worth: $1.1 billion
- Jack Schuler of Quidel Corp.-net worth: $1.1 billion
As of March 2021, the DOH and FDA have approved 133 brands of testing kits for commercial use in 279 testing labs nationwide.
READ: COVID test costs: Windfall for the rich, plague for the poor
Five of those approved testing brands were manufactured by billionaires from Sansure Biotech, Shenzhen New Industries Biological Engineering, Abbott Labs, Roche Diagnostics GmbH, and bioMérieux.
TSB
For more news about the novel coronavirus click here.
What you need to know about Coronavirus.
For more information on COVID-19, call the DOH Hotline: (02) 86517800 local 1149/1150.
The Inquirer Foundation supports our healthcare frontliners and is still accepting cash donations to be deposited at Banco de Oro (BDO) current account #007960018860 or donate through PayMaya using this link.