Villar chides BAI for letting private firm test ASF vaccines

Senator Cynthia Villar questions officials of the Bureau of Animal Industry on why it supposedly gave the job of clinical trials for ASF vaccines to a private firm when the bureau should be the one doing this. (Photo dated October 25, 2023, from Senate PRIB)
MANILA, Philippines — Senator Cynthia Villar grilled officials from the Bureau of Animal Industry (BAI) on Wednesday.
She accused them of allowing a private supplier to spearhead the nation’s clinical trials for the contentious African Swine Fever (ASF) vaccines from Vietnam.
Villar spoke at the Senate inquiry into the unauthorized sale of ASF vaccines in Philippine markets.
The probe was done by the committee on agriculture, food and agrarian reform headed by the senator.
The lawmaker asked BAI officials why they allowed private firm KPP Powers Commodities Incorporated to take over and conduct trials on 300,000 ASF jabs.
“Ang role namin is monitoring ‘yung kina-conduct na field trial. Hindi po kami ang actual na nagco-conduct,” BAI Director Paul Limson repeatedly clarified.
(Our role is monitoring the field trial being conducted. We are not the actual ones conducting.)
As Limson pointed out, BAI Assistant Director Arlene Vytiaco said the bureau did not hold a clinical trial on the vaccines.
She added KPP Powers Commodities Incorporated presented the field trial protocol to BAI.
Villar, however, insisted BAI “should not ask a private company to conduct a clinical trial,” stressing that the bureau should be the one doing it.
“In our system, it’s the private company. BAI is monitoring,” said Vytiaco in a mix of English and Filipino.
“Mali ang sistema. Clinical trial, the government should do it. Bakit niyo ia-allow ang private to do it?” the senator asked.
(The system is wrong. Clinical trials, the government should do it. Why will you allow the private to do it?)
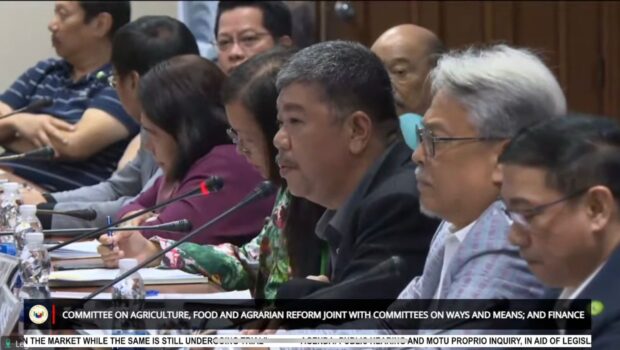
Bureau of Animal Industry Director Paul Limson (center, wearing black) responds to questions during the Senate hearing on the alleged unauthorized sale of African Swine Fever vaccines in the market. The probe was held on October 25, 2023. (Screengrab / Senate YouTube)
Later in the hearing, KPP Powers Commodities Incorporated Legal Counsel and Spokesperson Reynaldo Robles contradicted Limson and Vytiaco’s statements.
He clarified his firm’s participation.
He said they were not responsible for conducting trials on the controversial jabs.
“Hindi po kami nag conduct. Nag supply lang po kami ng vaccines,” Robles clarified.
(We did not conduct. We only supplied the vaccines.)
“Ang kumukuha ng samples ay may ari ng farm at ibinibigay sa BAI. Sila po ang nag test. ‘Yun ang nag monitor nila,” said Robles.
(The one who takes the samples is the farm owner and gives them to BAI. They are the ones who did the test. That’s what they monitored.)
“Lahat ng tests were done by BAI,” he stressed.
(BAI did all the tests.)
‘This should not be rushed’
For the farmers’ group Samahang Industriya ng Agrikultura (Sinag), the solution to the problem is simple: Wait and do not rush the inoculations.
“Dapat natuto tayo sa Covid experience eh,” said Sinag Executive Director Jason Cainglet.
(We should have learned from our Covid experience.)
“Yung Covid natin, naghintay tayo nang matagal to get the vaccine at merong approval ng World Health Organization,” he recalled.
(With Covid, we waited a long time to get the vaccine and have the approval of the World Health Organization.)
Cainglet said as a precautionary measure, the Philippines should let foreign countries purchase and use the vaccines first to determine if these injections are indeed effective.
“Very simple. It should not be rushed,” he added in Filipino.
RELATED STORIES:
USDA: ASF not done hurting pork production in PH