DOH: Guidelines for saliva-based RT-PCR test for COVID-19 out
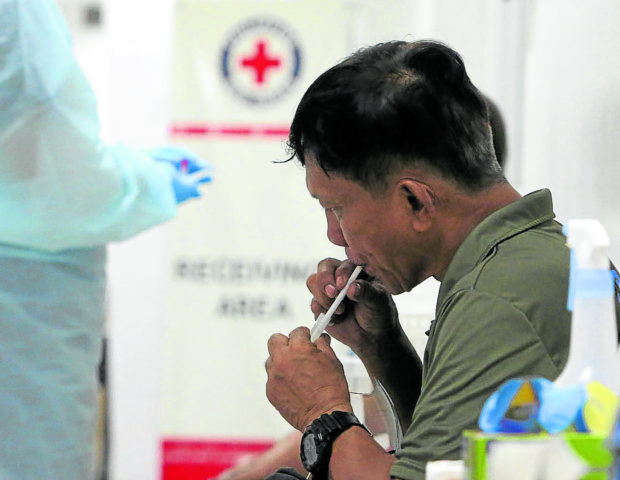
FILE PHOTO: A man spits in a small container using straw so his saliva can be collected and tested for the new coronavirus SARS-CoV-2, which causes COVID-19, at the Philippine Red Cross headquarters in Port Area, Manila, on January 26, 2021. INQUIRER/ MARIANNE BERMUDEZ
MANILA, Philippines — The guidelines on the use and administration of Saliva-based Reverse Transcription Polymerase Chain Reaction (RT-PCR) testing for COVID-19 have been released, the Department of Health (DOH) said Tuesday.
Under Department Memorandum (DM) No. 2021-0161, only licensed COVID-19 laboratories certified by the Research Institute for Tropical Medicine (RITM) are allowed to perform saliva-based RT-PCR.
It also said laboratories or facilities may only use test kits that have passed performance validation by the RITM or other RITM-recognized laboratories, as the kits should likewise have a special certification from the Food and Drug Administration.
“Point-of-care clinics, specimen collecting facilities, and disease reporting units can perform saliva specimen collection, provided that the specimen collectors have undergone training from the referral licensed COVID-19 laboratory certified to perform saliva testing.” the health agency said in a statement.
As for the collection of samples, the guidelines stipulate that morning saliva is preferred as a specimen due to higher viral load but this should not prevent specimen collection at any other time. It further noted that proper collection procedures should be strictly observed where patients should not eat, drink, brush their teeth, use mouthwash, or smoke for at least 30 minutes before sample collection.
Article continues after this advertisementDOH stressed that only trained saliva specimen collectors should administer the test and only medical doctors should interpret the results of these tests as results need to be correlated with the overall clinical and epidemiological context of the patient.
Article continues after this advertisementThe health agency, however, reiterated that nasopharyngeal and oropharyngeal swab specimens remain the gold standard for the diagnosis of COVID-19 through RT-PCR testing.
It said the conduct of saliva tests should only be done on certain conditions and are only deemed valid when such conditions are met.
In January, the national government approved the use of a cheaper, faster, safer saliva test for COVID-19 that will be initially conducted by the Philippine Red Cross.
RELATED STORIES
EXPLAINER: What is saliva-based Covid-19 test and where to get it
PH Red Cross completes study on COVID-19 saliva test — DOH
KGA
For more news about the novel coronavirus click here.
What you need to know about Coronavirus.
For more information on COVID-19, call the DOH Hotline: (02) 86517800 local 1149/1150.
The Inquirer Foundation supports our healthcare frontliners and is still accepting cash donations to be deposited at Banco de Oro (BDO) current account #007960018860 or donate through PayMaya using this link.